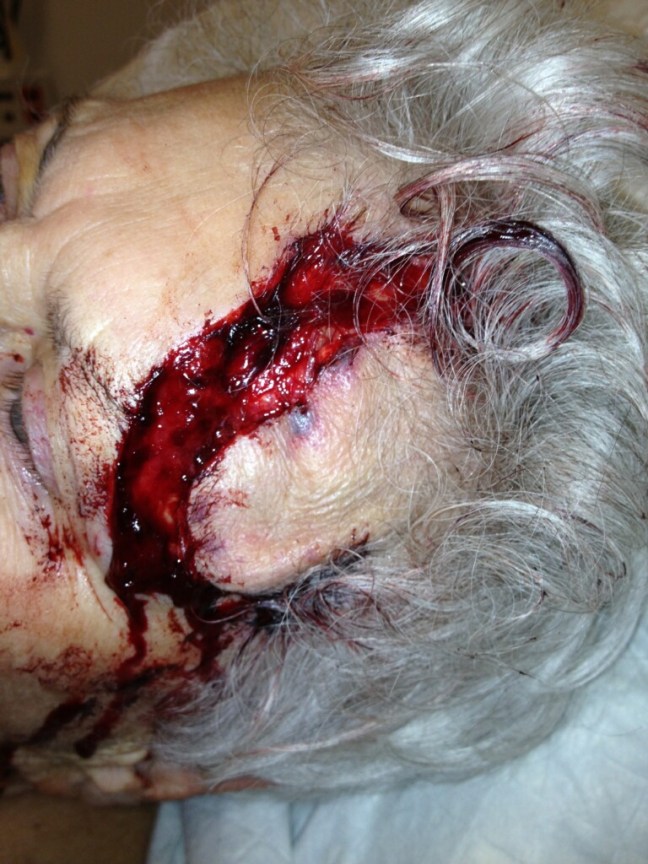
 Extensive forehead laceration, with large delay in presentation due to patient’s poor mobility.
Extensive forehead laceration, with large delay in presentation due to patient’s poor mobility.On a recent shift, I cared for an elderly patient with a fairly extensive and cosmetically deforming facial laceration. The patient was found down in her home, and due to inability to seek medical help was delayed in presenting to our ER for much longer than we would have liked. Upon evaluating the injury, I was fairly certain I wanted to suture it–or rather, I thought, were it my face, I would certainly want it repaired. However, a colleague on duty who saw the patient in passing shrugged and said, “Too bad you can’t do anything for that poor lady’s face. It happened 20 hours ago.”
By this comment, he was implying that the patient had come to see us outside of what’s commonly referred to as the “golden period”–in other words, the hours following an injury in which it’s considered safe to perform primary repair of a laceration.* This conversation got me thinking about the so-called golden period. Where did this concept come from? I know that along the path of my own training and experience, it’s been called in to question–but what’s the party line on what to do? And, what does the latest data show?
*Side note: Some would argue that we should halt this discussion right here: just refer the patient for delayed primary closure, or prescribe some antibiotics, and the problem is solved. Today’s discussion is for the rest of us, who may not have the luxury of close follow up with the patients we see. But, I do acknowledge the role for these strategies in some practice settings, and they are worthy of future blog discussions.
A Brief History of the “Golden Period”
The idea of the golden period is a pretty old one. It arises from (surprise surprise) cruel animal data. Way back in 1898, Professor Paul Leopold Friedrich lacerated the skin of guinea pigs, then inoculated the wounds with bacteria. He then determined that if the surrounding skin was not excised within 6 hours, the animals died.
…and there you have it: the initial determination of the “golden period.” Already, you can see the problems in relating this to your clinical practice. Artificially inoculating a wound with bacteria is not the same as the natural and (probably less pathogenic) bacterial exposure that occurs with traumatic lacerations; and excision of surrounding skin is not typically the standard practice for decontamination of wounds (though it is an option in certain situations–to be featured in an upcoming post!).
Still, it’s become pretty well accepted that there exists some window of time in which it is safe to primarily close traumatic lacerations. That window of opportunity is cited as anywhere between 3 to 24 hours, depending on what textbook or source you read. If you believe in the germ theory of disease, it’s a little hard to argue with the notion that the longer a wound festers, the more bacteria will colonize the wound, and the higher the infection risk becomes. So, I won’t attempt to argue the log-linear relationship between time since injury and bacterial colonization. But how that translates in to clinical risk of repair and absolute cut-off times for doing so is a different story.
Advancing the golden period (aka, is it a period or a “…” ?)
Research on this topic in the past few decades has sought to extend the golden period. To me, this implies that we all agree that the golden period is longer than 6 hours, we just don’t know exactly how much longer. Published research has determined safety in closing wounds up to 10 hours (Kanegaye 1997), 19 hours (Berk 1998) or even up to 24 hours old (Lammers 2003). The major flaws with these (and many other) studies–in no particular order– are lack of generalizability (single center, certain wound types only), the observational-only nature of the studies (granted, an RCT is tough to pull off here), and often in these studies evaluation of the golden period is only a secondary outcome (not the primary goal of the study). The American College of Emergency Physician’s Clinical Policy cites a period of 12 hours for traumatic lacerations of the extremities. It’s acknowledged even by ACEP to be way outdated (1999), though not yet updated.
Where we currently stand
So where does this jumbled data leave us today in terms of what to do in real-life clinical practice?
A meta-analysis published in Injury 2012 attempted to synthesize the existing, best quality data. While the methods of data extraction for the review were sound, the quality of the studies that met their criteria for inclusion were pretty low. The problems were many: even the included studies were only observational, the definition of the golden period varied (4-12 hrs), and the definition of “wound infection” varied. The upshot of the article was that no definite cut-off time for wound age (beyond which attempt at primary repair of the wound would be too high risk) could be defined.
Probably the largest problems in evaluating these studies is that there are far too many confounders. Wound age is just one of myriad variables that come in to play when it comes to determining infection risk. To name a few of the others: wound location, patient comorbidities, and trauma mechanism are all factors which make it very difficult to isolate time post-injury as a single variable. Or rather, one would have to conduct a very large study and collect data for a very long time to try to isolate the variable of interest.
But that’s okay, because this is the nature of real world emergency medicine. We consider these confounders every day in our assessment of traumatic lacerations, and let’s face it, this is how most of us really shape our decision making in regards to which wounds to close and which to leave alone.
I’ll leave you with this study to consider, published this year by Quinn, et al (after the afore-mentioned meta-analysis). In this observational study, the golden period (of 6 hrs) was examined, as well as multiple other factors, felt to be of potential relevance in predicting infection risk. The authors did find variables that were independent predictors for higher likelihood of infection: a history of diabetes, length of the laceration (>5cm), location of the wound, and level of contamination. Age of the laceration, interestingly, was not one of them.
Does this mean there was no increased risk to sewing my patient’s 20-hour-old forehead laceration? I highly doubt it. But, so many other factors needed to be accounted for simultaneously. The location was good and the contamination was low. She did not have diabetes or other immunocompromising conditions. The wound was long, which was a negative. But most importantly, the cosmetic appearance of her face was important to her. Once you’ve educated your patient on the factors which can affect healing of the wound, the best strategy should be to employ the concept of shared risk tolerance. She understood the risks, and accepted, and her wishes were concordant with my favored plan of care (were this a 20-hour-old laceration on the back of her knee, this may have been a different story). Thus, my–or rather, our–decision was made.
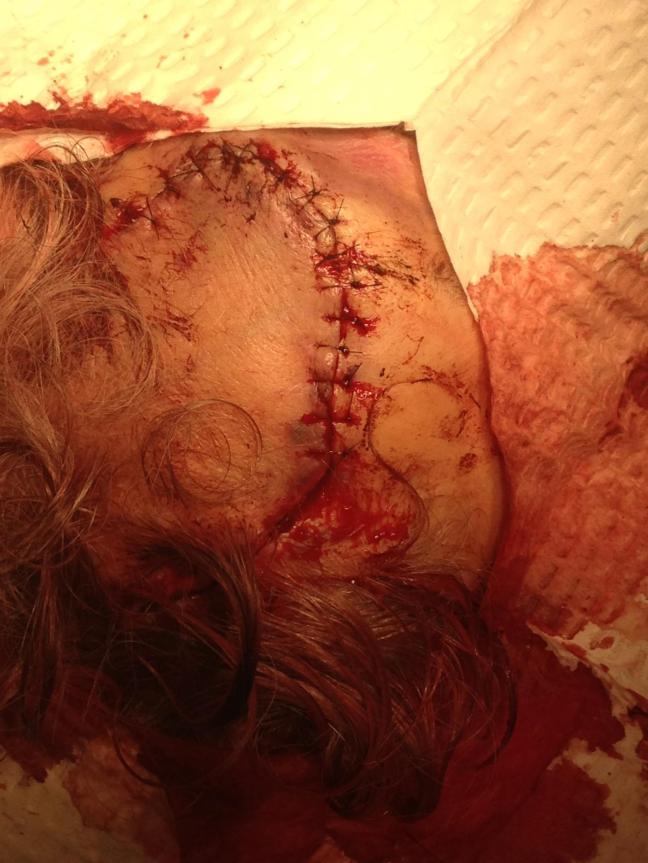 Forehead laceration, after meticulous repair with simple interrupted 6-0 nylon sutures.
Forehead laceration, after meticulous repair with simple interrupted 6-0 nylon sutures.The bottom line for me is, what distorts a research study as a “confounder” is what makes every patient, and every patient care situation, unique in real life. This is why we are needed as health care practitioners in the first place, and why our job can’t be carried out by automatons. It is our duty to be familiar with the myths so we can understand when they are a disservice to our patients; to stay up to date with what the best research data tells us; and then decide how to tailor it to each individual patient.



One thought on “The Golden Period”
Comments are closed.